Why Bacteriostatic Water is Essential for Pharmaceutical Research
Understand how this specialized sterile solution ensures the stability and purity of compounds in advanced scientific experiments.
The Foundational Chemistry of Bacteriostatic Water
In pharmaceutical research, the integrity of an entire study can hinge on the purity of a single solvent. A minor contamination can invalidate months of work, which is why seemingly simple components like diluents are held to exacting standards. Bacteriostatic water is one such critical component, designed specifically to maintain sterility over time.
At its core, bacteriostatic water is a sterile, nonpyrogenic water solution containing a specific preservative. This composition is strictly defined by pharmacopeial standards, such as the USP, which mandates the precise concentration of the bacteriostatic agent to ensure safety and efficacy. The key ingredient is 0.9% benzyl alcohol, which acts as a bacteriostatic agent. Its function is not to kill bacteria outright, but to inhibit their reproduction. Think of it as a security system that prevents intruders from multiplying, rather than eliminating them on sight.
This mechanism is what separates it from other solutions. While sterile water is free from microbes, it lacks a preservative and becomes susceptible to contamination after the first use. Bactericidal agents, on the other hand, actively kill bacteria. The use of benzyl alcohol as a preservative provides a middle ground, making the solution safe for repeated withdrawals from a single vial. This chemical property is fundamental to research integrity, as it prevents microbial growth and maintains compound stability for up to 28 days, ensuring reproducible results. Understanding these fundamentals is key to advancing research, a topic we explore further across our scientific discussions.
Critical Applications in Drug Development and Analysis
Moving from its chemical makeup to its practical function, bacteriostatic water is a workhorse in the modern laboratory. Its primary role is in reconstituting lyophilized compounds, the freeze-dried powders that represent a stable form for sensitive biologics, peptides, and other complex molecules. When you see a researcher carefully adding liquid to a vial of chalky powder, they are often bringing a potent compound back to life for an experiment.
This is particularly valuable in long-term peptide studies. The preservative allows a researcher to draw consistent, sterile doses from a single vial over several weeks. This avoids the laborious and error-prone process of preparing fresh solutions daily, which can introduce variability and compromise data. For researchers conducting these precise experiments, having a reliable source of reconstitution solution is paramount.
Beyond reconstitution, it serves as one of the most reliable pharmaceutical research diluents for creating precise concentrations. Whether for in-vitro assays or preclinical dose-response studies, accuracy is non-negotiable. Using a stable, preserved diluent ensures that the concentration prepared on day one remains the same on day twenty. This multi-dose capability delivers a powerful dual benefit. It is economical, reducing the waste associated with single-use vials. More importantly, it minimizes the risk of contamination that could derail an entire research project, making it an indispensable tool for ensuring experimental validity.
Protocols for Aseptic Handling and Storage
The effectiveness of bacteriostatic water depends entirely on proper handling. Even with a preservative, poor technique can introduce contamination and degrade sensitive compounds. Adhering to strict aseptic protocols is not just best practice; it is essential for reproducible science.
Every withdrawal from a multi-dose vial must follow a standard procedure to maintain sterility. The process is straightforward but requires discipline:
- Disinfect the Vial Septum: Before every use, vigorously wipe the rubber stopper with a sterile 70% alcohol swab and allow it to air dry completely. This simple step removes surface contaminants.
- Use a New Sterile Needle and Syringe: Never reuse a needle or syringe, even for the same vial. Microscopic contamination on a used needle can compromise the entire solution.
- Draw the Solution Correctly: To avoid creating a vacuum, first draw a volume of air into the syringe equal to the dose you intend to withdraw. Inject the air into the vial before drawing the solution.
Once opened, storage conditions are just as critical. The vial must be refrigerated at 2°C to 8°C (36°F to 46°F) and discarded 28 days after the first use, regardless of how much solution remains. Common errors like storing a reconstituted peptide at room temperature or failing to swab the vial can lead to rapid bacterial growth and compound degradation, skewing results. To prevent this, always label the vial with the date it was first opened and the calculated discard date. This simple act of documentation is a cornerstone of laboratory safety and data integrity.
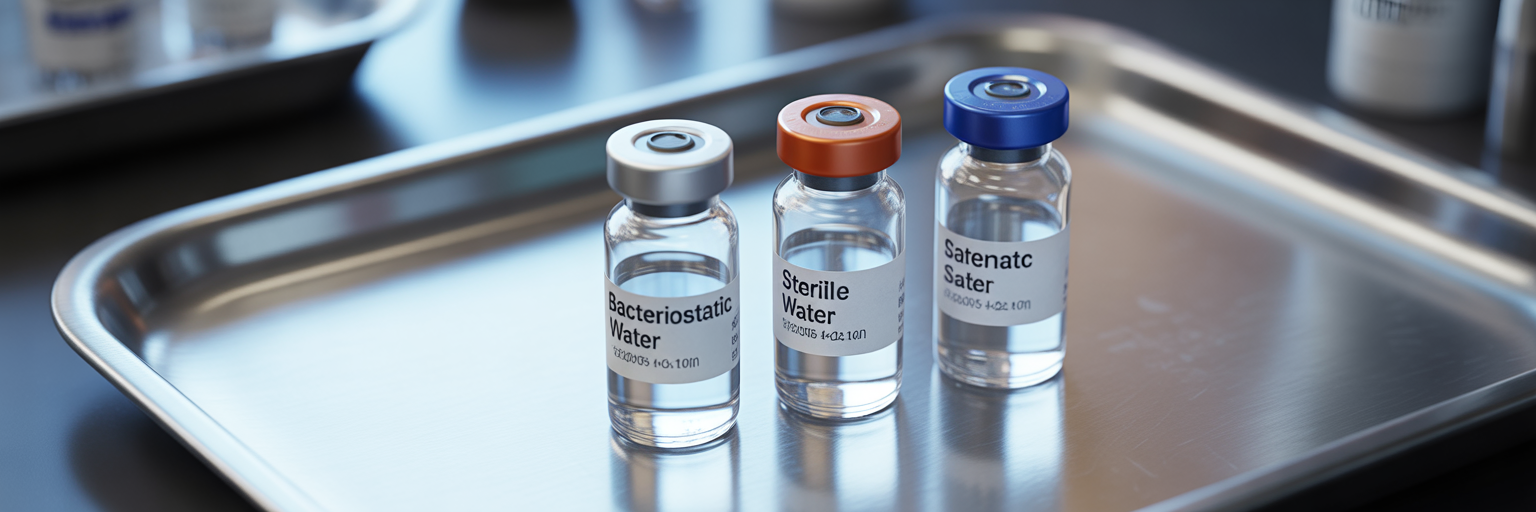
Distinguishing Between Common Sterile Diluents
Choosing the correct diluent is a critical decision that depends on the compound, the intended use, and the required shelf-life after opening. While bacteriostatic water is versatile, it is not a one-size-fits-all solution. Understanding the differences between it and other common diluents helps researchers avoid costly errors.
The most frequent point of confusion in the sterile water vs bacteriostatic water debate is the preservative. Sterile Water for Injection (SWFI) is pure, sterile water with no additives. Its lack of a preservative means it is strictly for single-dose use. Once opened, any unused portion must be discarded immediately to prevent microbial contamination. It is the diluent of choice when benzyl alcohol is contraindicated for a specific compound or patient.
Normal Saline (0.9% NaCl) introduces another variable: tonicity. This isotonic solution matches the salt concentration of human blood, making it the standard for intravenous (IV) injections to prevent red blood cell damage. Bacteriostatic water, being hypotonic, is typically reserved for intramuscular (IM) or subcutaneous (SC) injections where tonicity is less of a concern. The distinctions between these preparations are codified in pharmaceutical standards, which outline their specific formulations and intended applications. Researchers must always check a compound’s monograph, as some molecules are incompatible with benzyl alcohol and require a specific diluent. Choosing the correct diluent is a critical step, and a range of pharmaceutical-grade solutions are available to meet specific research needs.
| Diluent | Composition | Primary Use Case | Shelf-Life After Opening |
|---|---|---|---|
| Bacteriostatic Water | Sterile water + 0.9% Benzyl Alcohol | Multi-dose reconstitution for IM/SC injection | Up to 28 days |
| Sterile Water for Injection (SWFI) | Sterile water only (no preservative) | Single-dose reconstitution; when benzyl alcohol is contraindicated | Discard immediately |
| Normal Saline (0.9% NaCl) | Sterile water + 0.9% Sodium Chloride | Isotonic diluent for IV injections; wound cleaning | Single-dose unless preserved |
Note: This table provides a general guide. Researchers must always consult the specific drug or peptide’s monograph for the required diluent to ensure compound stability and patient safety.
Regulatory Compliance and Critical Safety Measures
The reliability of bacteriostatic water is built on a foundation of stringent regulatory oversight. Manufacturers must adhere to pharmacopeial standards, such as those from the USP and European Pharmacopoeia (EP), which guarantee sterility, purity, and the precise 0.9% concentration of benzyl alcohol. These are not guidelines; they are non-negotiable requirements for any solution intended for injection.
However, the most critical safety measure is a stark warning: bacteriostatic water is absolutely contraindicated for use in neonates. This is due to the presence of benzyl alcohol. Newborns, particularly premature infants, lack the enzymes to metabolize it, leading to a fatal toxic reaction known as “gasping syndrome.” This critical contraindication is explicitly stated on product labeling, as highlighted in guidelines from regulatory bodies and manufacturers, such as this example from Pfizer’s product labeling. There are no exceptions to this rule.
For this reason, researchers and clinicians have an ethical and professional responsibility to verify the label of every vial before use. Key information includes:
- The full product name: bacteriostatic water for injection
- Preservative content and concentration
- A valid expiration date and lot number
- The explicit warning: “Not for use in newborns”
Finally, any solution that is expired, has been stored improperly, or appears cloudy or discolored must be discarded immediately. Using a compromised diluent not only invalidates research data but also poses a direct safety risk. Upholding these standards is fundamental to responsible science.
The Future Role in Advanced Therapeutic Research
While bacteriostatic water may seem like a basic laboratory supply, its role in pharmaceutical innovation is more relevant than ever. As research pushes into biologics, custom peptides, and personalized medicine, the need for stable, reliable reconstitution methods becomes even more critical. These complex molecules are often produced in small, expensive batches, making the multi-dose capability of bacteriostatic water essential for minimizing waste and ensuring consistency.
Looking ahead, its principles are influencing the design of novel drug delivery systems. Multi-dose auto-injectors for chronic conditions and sustained-release formulations both depend on maintaining sterility long after the first use. The simple, effective mechanism of bacteriostatic water provides a proven model for these advanced applications. While the scientific community continues to explore alternative bacteriostatic agents, the reliability and long-standing safety profile of benzyl alcohol (outside of neonatal use) solidify its position as the current standard.
Ultimately, its value lies in its simplicity and dependability. It is an unassuming but indispensable tool that allows researchers to focus on discovery, confident in the stability and sterility of their compounds. It is a foundational element that quietly supports the entire research pipeline, from early-stage experiments to preclinical trials. As research evolves, so do the tools that support it, reflecting our commitment to scientific advancement.